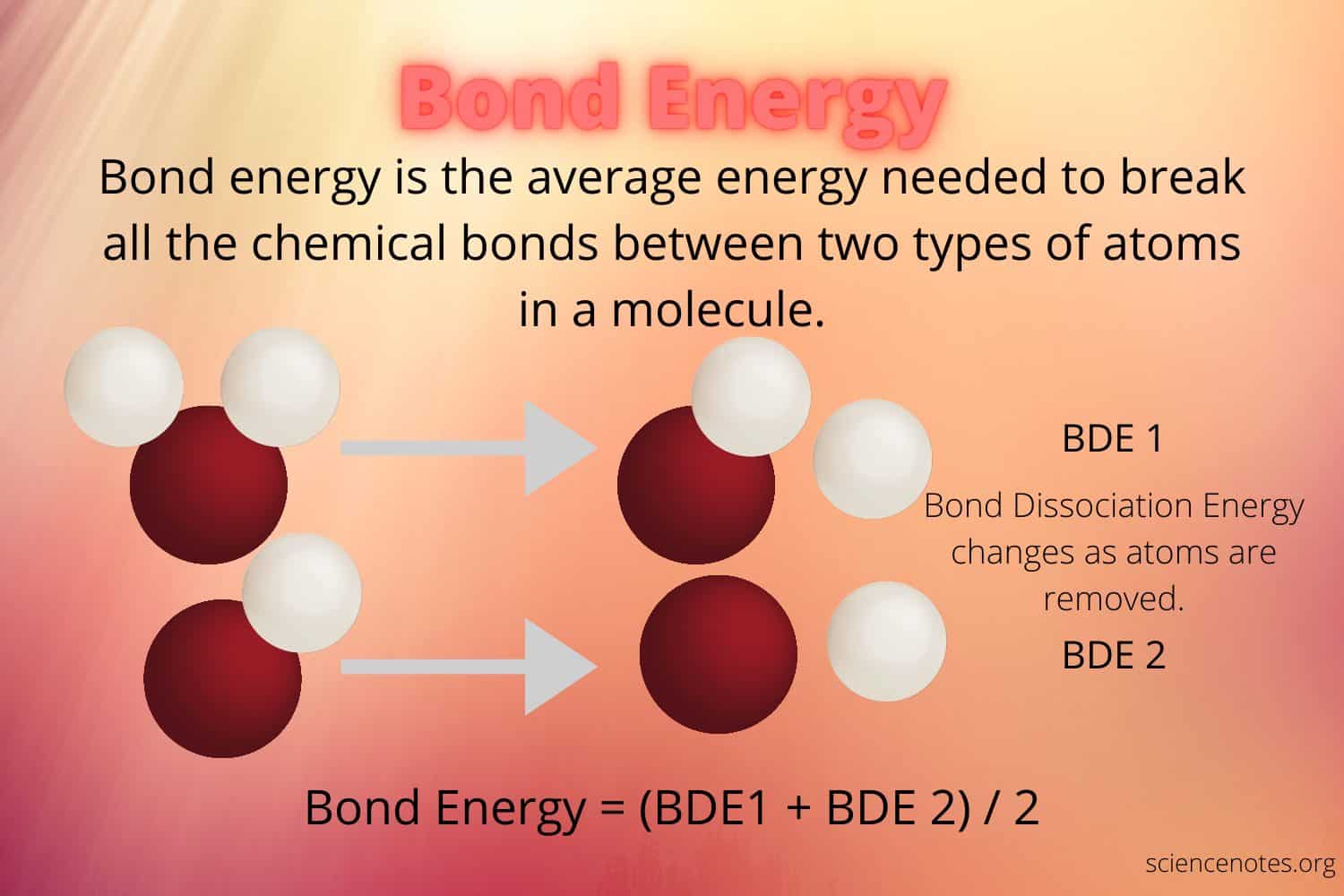
Table of Contents
Preparing For a Gas Laws Quiz
If you’re a high school student, you might be taking a gas laws quiz. These questions are meant to test your understanding of the concepts. You will find problems on pressure, volume, temperature, and the Ideal Gas Law. Make sure you know these laws before you take the quiz. If you don’t, you might be surprised by how difficult they are! To prepare for this exam, review the answers and find out where you need additional help.

Gas laws are important, especially for those studying the physical sciences. Learn the concepts of these laws by taking a quiz. The most important one to know is Boyle’s law, which defines a gas’s ideal state. Another one is Charles’s law, which defines the amount of moles in a sample. Using these laws, you can figure out the volume of gas in a given space. Then, you can use the Ideal Gas’s ideal state and calculate the temperature of the ideal gas.
Lastly, you should know the Ideal Gas Law. This law describes the physical behavior of ideal gases. It includes temperature, volume, and number of moles. The constant, “R,” is dependent on the units of pressure, and this quiz will test your knowledge of the ideal gas’ ideal state. Hopefully, this quiz will help you pass your next class! And, don’t forget to use the practice quiz to review the material before your final.
It’s important to know that the Ideal Gas Law is a formula used to determine the pressure of a gas. The equation is based on the mean free path, which is the state of a perfect gas. The ideal gas has a value of 2.66 x 1023. This equation also applies to the pressure of a substance when it’s in a cylinder. If it’s not a real gas, you can substitute a liquid or a solid at the end of the cylinder.
The Ideal Gas Law describes the state of an ideal gas. The ideal gas occupies a volume V and a temperature T. It also has constant pressure. Its temperature will decrease to T/2 if the pressure is equal to the temperature of an ideal gas. However, it doesn’t have this property. Despite these properties, it’s still impossible to make an ideal gas. The underlying theory explains the behavior of an atom.
You can study the Ideal Gas Law in your online course by taking a gas laws quiz. This quiz will test your understanding of the Ideal Gas Law. If you’ve mastered the material in class, you can take a quiz based on it. The test will give you the opportunity to check your knowledge of the topic. A good chance in your exam will be to have some questions that relate to the concepts you’ve learned. For example, a question about pressure and volume will depend on the pressure.
The ideal gas law can be formulated with the help of flashcards. You can also use games to practice. In the ideal gas, the temperature of a gas is constant. This is the same as that of an ideal fluid. The two ideal gases are equivalent. They have different volumes, but they all have the same temperatures. A great example of an ideal gas is a fluid that can be heated to a high temperature and has a constant volume.
The ideal gas occupies a volume V at a constant pressure. This gas is ideal because its volume is constant, while the ideal gas has a high temperature. As a result, its temperature will decrease by half. This is the perfect example of a fluid’s ideal state. A liquid is the ideal state of a substance. It is also an example of a fluid that has the potential to melt. Its volume is the quantity of energy it can carry without breaking.
The ideal gas law describes the ideal gas’s state at a given temperature. If you’ve ever seen an ideal gas, then you’ve probably seen one. The ideal gas law is the inverse of the temperature of the ideal gas. This law states that the pressure of an ideal gas is always 2.66 x 1023 mol. The temperature of a material that has the perfect volume is called the absolute zero. If the pressure is lower than the volume of the liquid, the volume is smaller than the amount of the molecule.