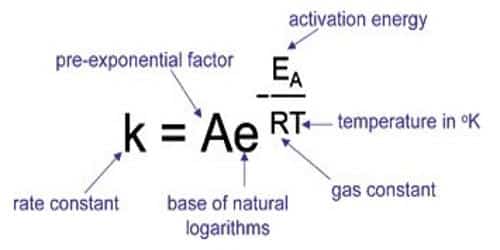
Arrhenius Formula
1
) The price consistent for the first-order dehydration of tert-butyl alcohol at 500oC is 1.20 x 10-4 s-1. The price consistent for this procedure at 600oC is 6.80 x 10-3 s-1. Compute the activation power for this response.
( a) -227 kJ/mol
( b) 227 kJ/mol
( c) 318 kJ/mol
( d) 100. kJ/mol
(e) -100. kJ/mol
2
) Use the chart to the right to determine the price continuous for the complying with response at 40.0 C. 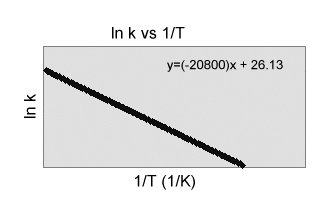
CH 3 CH 2 NO 2(aq) C 2 H 2
( g) +HNO 2( aq )(a)3.1 x 10 -18 sec -1
( b) 9.56 x 10-15 sec-1
( c) -8.3 x 105 sec-1
(d) -6.5 x 106 sec-1
3
) The activation power for the isomerization of cyclopropane to propene is 274 kJ/mol. By what variable does the price of response rise as the temperature level increases from 500oC to 550oC?
( a) 1.0
( b) 13
( c) 2.6
( d) 4.0 x 102
4
) The price consistent for a particular first-order response at 27oC is 4.9 x 10-4 s -1 as well as is 0.18 s-1 at 227oC. Calculate the activation power for this response.
( a) 8.55 x 104 kJ/mol
( b) 9.80 x 104 kJ/mol
( c) 3.68 x 104 kJ/mol
( d) -1.50 x 103 kJ/mol
(e) 1.50 x 103 kJ/mol
5
) Use the chart to the right to determine the price continuous for the adhering to response at 40.0 C. 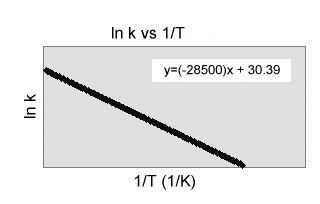
C 2 H 5 Cl(aq) C 2 H 4(g)+HCl(aq)(a)-9.29 x 10 6 sec -1(b)-1.51 x 106sec -1(c)
-57.0 sec -1(d) 1.70x 10 -25 sec -1
6 ) The response price triples when the temperature level raises from 15 o C to 35 o C for a certain response. What is the activation power for this response? (a) 2.39 x 102 J/mol(b )2.06 x 10 -3
J/mol(
c)7.92 x 10 -2 J/mol(d )4.05 x 10 4 J/mol House Online Assignments