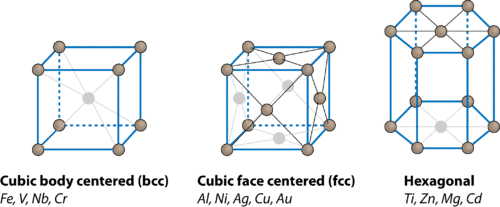
Crystal Frameworks
1
) Nickel has a system cell that is a face-centered cubic with a quantity of 43.763 Å3. The atom at the facility of each face simply touches the atoms at the edges. What is the atomic distance of nickel?
(a) 0.881 Å
(b) 1.246 Å
(c) 2.492 Å
(d) 1.661 Å
(e) 0.831 Å
2
) Cadmium oxide has a device cell that is a face-centered cubic. The ionic span of Cd2+ is 0.97 Å and also the ionic distance of O2- is 1.32 Å. What is the thickness of CdO?
(a) 1.3 x 1024 g/cm3
(b) 8.9 g/cm3
(c) 2.2 g/cm3
(d) 8.9 x 10-24 g/cm3
(e) 13 g/cm3
3
) Which of these materials has the most affordable melting factor?
(a) LiBr
(b) CaO
(c) CARBON MONOXIDE
(d) CH3OH
4
) Cesium chloride has a system cell that is a body-centered cubic and also salt chloride has a device cell that is a face-centered cubic. Which of the complying with represent this distinction?
(a) the reduced electronegativity of the cesium atom
(b) the bigger distance of the cesium ion
(c) the greater cost on the cesium center
(d) the reduced ionization power of the valence electrons discovered in cesium
5
) Which of the complying with is one of the most vital aspect influencing the device cell framework of an ionic substance?
(a) the distance proportion of the ions
(b) the atomic mass of the ions
(c) the cost on the ions
(d) the price at which condensation takes place
6
) Palladium has a system cell that is a face-centered cubic. If the thickness of palladium is 12.0 g/cm3 at 25 ° C, what is its distance?
(a) 0.107 nm
(b) 138 pm
(c) 152 pm
(d) 1.96 x 10-8 nm
(e) 1.96 x 10-8 centimeters
7
) The size of a system cell for an atom of gold is 407.9 pm. If the thickness of gold is 19.3 g/cm3, what kind of cubic latticework does gold have?
(a) basic or primitive cubic
( b) body-centered cubic
( c) face-centered cubic
8
) Silver has a device cell that is a face-centered cubic as well as a thickness of 10.50 g/cm3. Utilizing this information, compute Avogadro’s number.
( a) 6.02 x 1023
House
|
|