Boyle’s Regulation
Boyle’s
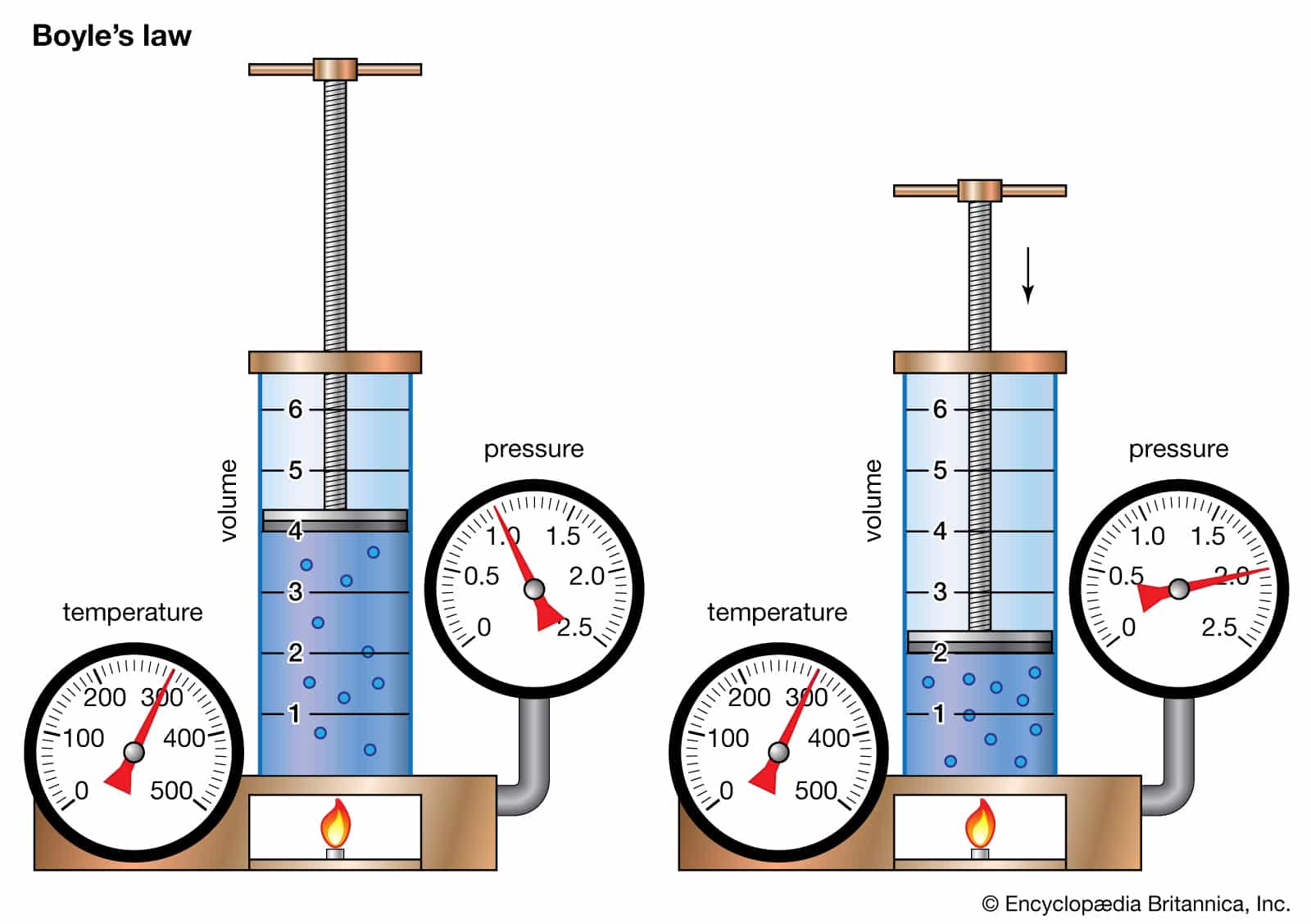
Law mentions the partnership in between the quantity as well as stress of a gas as the temperature level as well as the variety of moles continue to be continuous. It specifies that the quantity and also stress are vice versa symmetrical.
If the quantity is raised on a gas the stress will certainly reduce and also as the quantity is lowered the stress will certainly raise. The very first chart reveals the inverted partnership in between quantity and also stress. Mathematically mentioned, Boyle’s Law claims that PV = k, where k is a consistent for a provided quantity of gas at consistent temperature level.
The chart listed below programs the direct connection when V is outlined versus 1/P.
Home Chart Gallery