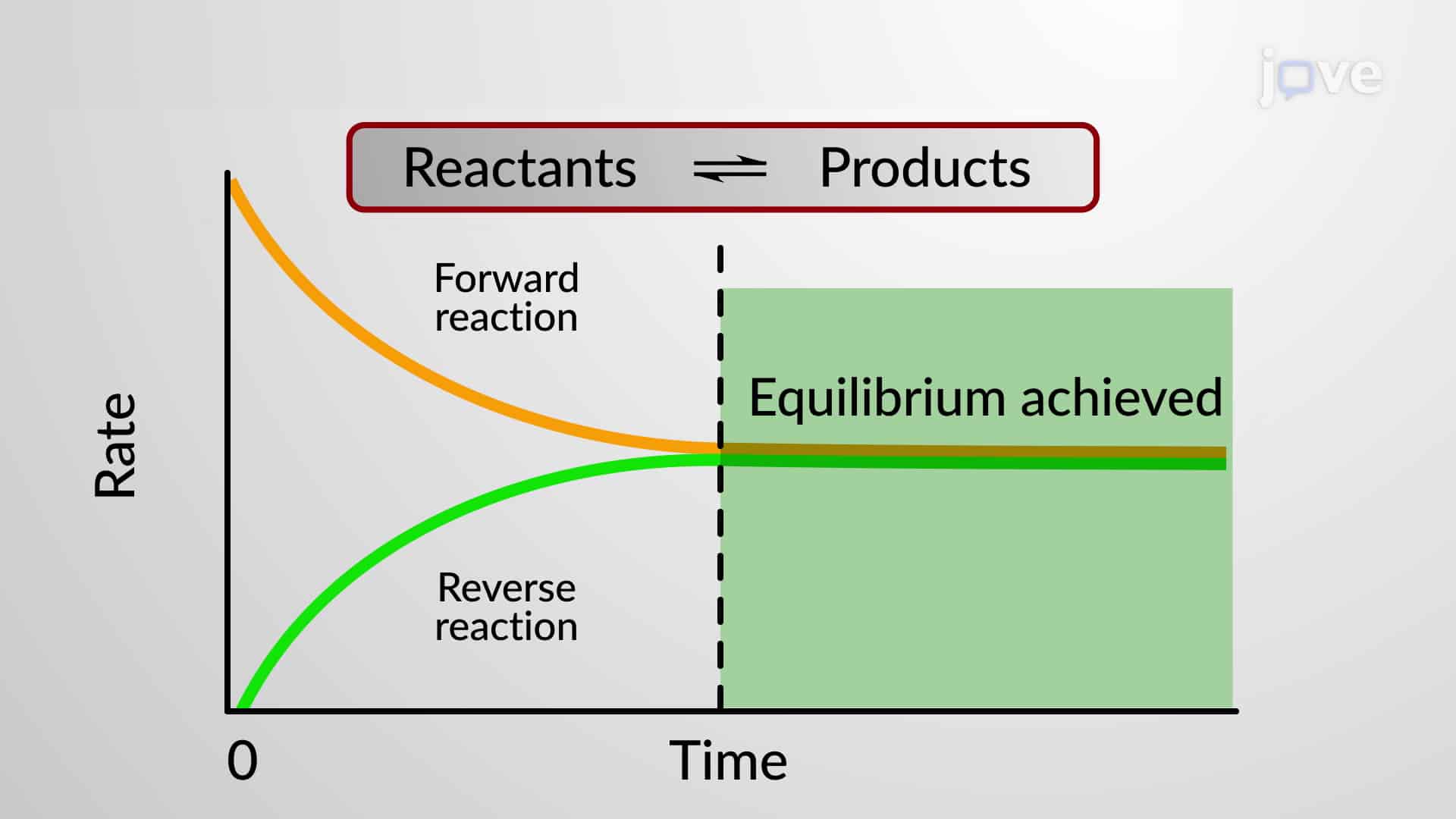
Chemical Equilibrium
The complying with troubles are extracted from T. L. Brown, H. E. Lemay, B. E. Bursten, & J. R. Burdge, Chemistry: The Central Science, Ninth Edition, Chapter 15. To see the option to an issue or to return from the option, click on the variety of the trouble. 12 )The stability consistent for the response
2NO(g)+Br 2(g)2NOBr(g)is K eq=1.3 x 10 -2 at 1000 K.(a) Calculate K eq for 2NOBr(
g )2NO(g)+Br 2 (g)(b)At this temperature level does the balance support NO and also Br 2, or does it prefer NOBr? 14) Think about thecomplying with balance, for which K eq=0.0752 at 480 °
C:
2Cl 2(g
![]() ? < img elevation= "16 "src= "https://mmsphyschem.com/wp-content/uploads/2017/06/darrow-7323390.gif"size =”33″>(b)What is the worth
? < img elevation= "16 "src= "https://mmsphyschem.com/wp-content/uploads/2017/06/darrow-7323390.gif"size =”33″>(b)What is the worth
![]() of K eq for the response Cl 2(g)+H 2 O( g)2HCl(g)+1/2O 2(g)?(c)What is the worth of K eq for the response 2HCl(g)+1/2O 2(g) Cl 2( g) +H 2 O(g)? 22 )Phosphorus trichloride gas as well as chlorine gas respond todevelop phosphorus pentachloride gas: PCl 3(g)+Cl 2(g )PCl 5(g)A gas vessel is billed with a combination of PCl 3 (g)and also Cl 2(g), which is enabled to equilibrate at 450 K. At stability the partial stress of the 3 gases are P PCl3=0.124 atm machine, P Cl2 = 0.157 atm machine, and also P PCl5=0.130 atm machine.(a)
of K eq for the response Cl 2(g)+H 2 O( g)2HCl(g)+1/2O 2(g)?(c)What is the worth of K eq for the response 2HCl(g)+1/2O 2(g) Cl 2( g) +H 2 O(g)? 22 )Phosphorus trichloride gas as well as chlorine gas respond todevelop phosphorus pentachloride gas: PCl 3(g)+Cl 2(g )PCl 5(g)A gas vessel is billed with a combination of PCl 3 (g)and also Cl 2(g), which is enabled to equilibrate at 450 K. At stability the partial stress of the 3 gases are P PCl3=0.124 atm machine, P Cl2 = 0.157 atm machine, and also P PCl5=0.130 atm machine.(a)
What is the worth of K eqat this temperature level?(b)Does the stability support catalysts or items? 24)A combination of 1.374 g of H 2 as well as 70.31 g of Br 2 is warmed in a 2.00-L vessel at 700 K. These materials respond as adheres to: H 2(g)+Br 2(g) 2HBr( g)At balance the vessel is located
![]() adhering to response has K eq=0.345: 2SO 2(
adhering to response has K eq=0.345: 2SO 2(
g ) +O 2(g)2SO 3(g )In a balance blend the partial stress of SO 2and also O 2 are 0.165 atm machine
and also 0.755 atm machine, specifically.
What
isthe balance partial stress of SO 3 in the combination? 38 )At 21.8 ° C,
![]() K eq=7.0 x 10 -2 for thebalance NH 4 HS(s )NH 3(g )+H 2 S (g)Calculate the balance partial stress of NH3 as well as H 2 S if an example of strong NH 4 HS
K eq=7.0 x 10 -2 for thebalance NH 4 HS(s )NH 3(g )+H 2 S (g)Calculate the balance partial stress of NH3 as well as H 2 S if an example of strong NH 4 HS
is
putin a shut vessel as well as disintegrates till balance is gotten to. House Chem II AP Helpers